HAEM4Backup:Acute Myeloid Leukemia (AML) with t(8;21)(q22;q22.1); RUNX1-RUNX1T1
Primary Author(s)*
Christine Bryke, MD
Beth Israel Deaconess Medical Center, Boston, MA
Cancer Category/Type
Acute myeloid leukemia
Cancer Sub-Classification / Subtype
Acute myeloid leukaemia (AML) with t(8;21)(q22;q22.1) resulting in RUNX1-RUNX1T1 fusion
Definition / Description of Disease
Acute myeloid leukemia (AML) with t(8;21)(q22;q22) resulting in RUNX1-RUNX1T1 gene fusion defines a subtype of AML with neutrophil lineage maturation. In the 2016 revision to the World Health Organization (WHO) classification of myeloid neoplasms and acute leukemia, it is in the group of AML with recurrent genetic abnormalities[1]. In the older French-American-British (FAB) classification system, t(8;21)(q22;q22) positive AML belonged to the acute myeloblastic leukemia with maturation M2 category and accounted for 10% of cases. t(8;21)(q22;q22) can occasionally be seen in therapy-related AML. In those instances the leukemia is not classified in the group of AML with recurrent genetic abnormalities due to clinical and prognostic differences. AML with t(8;21)(q22;q22) is a core binding leukemia (see Genes and Main Pathways Involved section below)[2].
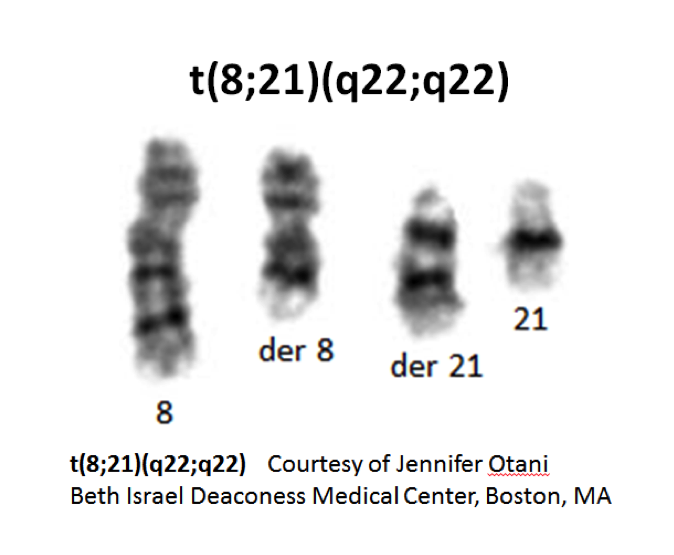
- t(8;21)(q22;q22) is a translocation involving the long arms of chromosomes 8 and 21 that defines a distinctive cytogenetic subtype of acute myeloid leukemia.
- Complex translocations involving chromosomes 8, 21 and a third chromosome occur; the derivative chromosome 8 in the t(8;21)(q22;q22) and the variant t(8;21;var) carries the RUNX1-RUNX1T1 gene rearrangement.
- Cases with cryptic RUNX1-RUNX1T1 gene rearrangement have been reported.
- Note: The RUNX1T1 gene is currently located at 8q21.3. This gene was previously located at 8q22, which explains the discrepancy in the breakpoint given in the name for this WHO-defined disease entity.
Synonyms / Terminology
AML1-ETO
Epidemiology / Prevalence
Approximately 1-5% of AML cases have the t(8;21)(q22;q22.1)
- Occurs mainly in younger patients
- Mean age is 30 years
- Most common type of AML in children
Clinical Features
- May present as a myeloid sarcoma and, if so, the bone marrow aspirate blast count may be misleadingly low
- Concurrent systemic mastocytosis has been reported.
Sites of Involvement
Bone marrow
Morphologic Features
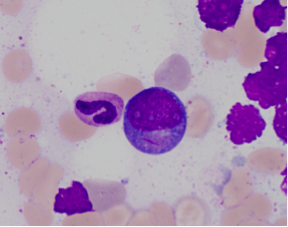
- Large blasts with abundant basophilic cytoplasm often with azurophilic granules and perinuclear clearing
- Long sharp Auer rods are frequently observed.
- Promyelocytes, myelocytes and metamyelocytes may have abnormal nuclear segmentation and cytoplasmic staining.
- Normal eosinophil precursors are frequently increased and do not show the nuclear and cytoplasmic abnormalities seen in AML with inv(16)(p13.1q22) or t(16;16)(p13.1;q22).
- Cases with t(8;21)(q22;q22.1) and less than 20% blast percentage in the marrow should be classified as AML rather than myelodysplastic syndrome according to the WHO 2017[3].
Immunophenotype
- Blasts express CD34, HLA-DR, MPO, CD13, CD33 (weak), CD15 and/or CD65, co-expression of CD34. Lymphoid markers: CD19, PAX5, sometimes cytoplasmic CD79a
- Occasional weak TdT positivity and/or CD56 expression may have adverse prognosis
| Finding | Marker |
|---|---|
| Positive (universal) | CD34, HLA-DR, MPO, CD13, CD33 (weak), TdT (weak), CD56 (fraction of cases) |
| Positive (subset) | CD15 and/or CD65 |
| Positive (Lymphoid markers) | CD19, PAX5, sometimes cytoplasmic CD79a |
| Negative (universal) | |
| Negative (subset) |
Chromosomal Rearrangements (Gene Fusions)
| Chromosomal Rearrangement | Genes in Fusion (5’ or 3’ Segments) | Pathogenic Derivative | Prevalence |
|---|---|---|---|
| t(8;21)(q22;q22.1) | 5' RUNX1 / 3'RUNX1T1 | der(8) | 1- 5% of AML |
Characteristic Chromosomal Aberrations / Patterns
Additional aberrations in >70% of cases with t(8;21); common loss of a sex chromosome or deletion 9q
Genomic Gain/Loss/LOH
9q22 loss
Gene Mutations (SNV/INDEL)
AML with t(8;21)(q22;q22) has genetic heterogeneity at the molecular level. Several genes have been identified that are frequently mutated in this subset of AML, some of them adversely affecting prognosis.
- KIT mutations occur in 20-25% of cases and are associated with a high risk of relapse in AML with t(8;21)(q22;q22)[4]. Patients with KIT mutations have a significantly shorter overall survival and disease free survival periods than those without KIT mutations[5].
- In a study of 139 t(8;21)(q22;q22) AML patients were investigated for possible mutations in KIT and 10 other genes (ASXL1, FLT3, NPM1, MLL, IDH1, IDH2, KRAS, NRAS, CBL, and JAK2)[6]. 49.6% had one mutation in addition to the t(8;21)(q22;q22). These were most often KIT, NRAS and ASXL1. 16.5% of patients had ≥2 mutations. Infrequent accompanying mutated genes found in only 2.9-5% of cases include in decreasing frequency: FLT3-ITD, FLT3-TKD, CBL, and KRAS, IDH2 and JAK2. These mutations did not adversely affect prognosis. NPM1 mutations are thought to be mutually exclusive of RUNX1-RUNX1T1.
- ASXL2 mutations have been identified in greater than 20% of patients with t(8;21), but not in patients with inv(16)/t(16;16) or RUNX1-mutated AML[7]. ASXL2 mutations are similarly frequent in adults and children with t(8;21) and are mutually exclusive with ASXL1 mutations. Although overall survival is similar between ASXL1 and ASXL2 mutant t(8;21) AML patients and their wild-type counterparts, patients with ASXL1 or ASXL2 mutations had a cumulative incidence of relapse that is significantly higher than with their ASXL1 or ASXL2 wild-type counterparts[7].
- Recurring ZBTB7A mutations have been identified in 23% (13/56) of AML t(8;21) patients, including missense and truncating mutations resulting in alteration or loss of the C-terminal zinc-finger domain of ZBTB7A[8]. The transcription factor ZBTB7A is important for hematopoietic lineage fate decisions and for regulation of glycolysis. On a functional level, it has been shown that ZBTB7A mutations disrupt the transcriptional repressor potential and the anti-proliferative effect of ZBTB7A. The specific association of ZBTB7A mutations with t(8;21) rearranged AML points towards leukemogenic cooperativity between mutant ZBTB7A and the RUNX1/RUNX1T1 fusion[8].
- WT1 mutations have been found to be relatively common (13.8%) in core binding factor leukemia[9]. Low WT1 transcript levels at diagnosis predicted poor outcomes of acute myeloid leukemia patients with t(8;21) who received chemotherapy or allogeneic hematopoietic stem cell transplantation[10].
| Gene | Mutation | Oncogene/Tumor Suppressor/Other | Presumed Mechanism (LOF/GOF/Other; Driver/Passenger) | Prevalence (COSMIC/TCGA/Other) |
|---|---|---|---|---|
| KIT | 20-30% |
Other Mutations
| Type | Gene/Region/Other |
|---|---|
| Concomitant Mutations | NRAS and ASXL1 |
| Secondary Mutations | |
| Mutually Exclusive | ASXL1 and ASXL2 |
Epigenomics (Methylation)
None
Genes and Main Pathways Involved
Genes:
- RUNX1T1 (also known as ETO) on 8q22 is transcribed from telomere to centromere; 3 proline rich domains; 2 zinc finger, and a PEST region in the C-terminus; tissue restricted expression; nuclear localization; putative transcription factor.
- RUNX1 (also known as AML1 and CBFA) on 21q22; contains a runt domain and a transactivation domain in the C-terminus; forms heterodimers; widely expressed; nuclear localization; transcription factor (activator) for various hematopoietic-specific genes.
DNA:
- t(8;21)(q22;q22) results in a 5’RUNX1-3’RUNX1T1 hybrid gene involving fusion of the very 5’ end of the RUNX1T1 gene to RUNX1 in an ~25 kb breakpoint region between exons 5 and 6.
Protein:
- A fusion protein with the N-terminus runt domain (responsible for DNA binding and protein-protein interaction) of RUNX1 fused to the 577 C-terminal residues of RUNX1T1. The reciprocal product is not detected. The fusion protein retains the ability to recognize the RUNX1 consensus DNA binding site (it is a negative dominant competitor with the normal RUNX1) and to dimerize with the CBFβ subunit in the heterodimeric core binding factor. The core binding factor is a transcription factor that plays an essential role in regulation of normal hematopoiesis. It is composed of a DNA-binding CBFα (RUNX1) chain and a non-DNA-binding CBFβ chain. It is likely oncogenic due to altered transcriptional regulation of normal RUNX1 target genes. Animal studies suggest that the fusion proteins alone are not able to induce leukemia and that additional genetic alterations are required for leukemogenic transformation[11][12].
Key cellular pathways:
- The normal transcriptional activity of the RUNX1 protein in the core binding factor is affected in a negative manner by the chimeric protein encoded by the RUNX1-RUNX1T1 fusion gene. It has been proposed that the RUNX1-RUNX1T1 protein recruits histone deacetylases and DNA methyl transferase 1 to core binding factor target genes[13]. This leads to increased chromatin deacetylation and promotor hypermethylation, resulting in gene transcriptional repression and disruption of normal cellular pathways of hematopoiesis.
Diagnostic Testing Methods
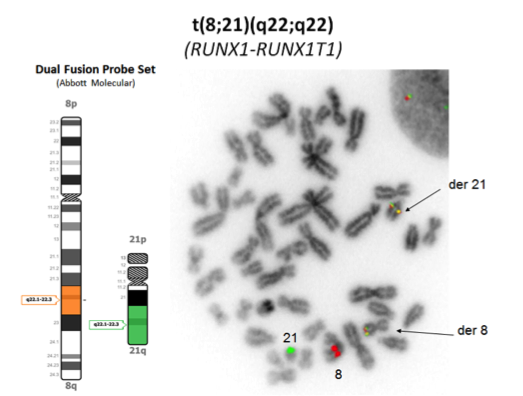
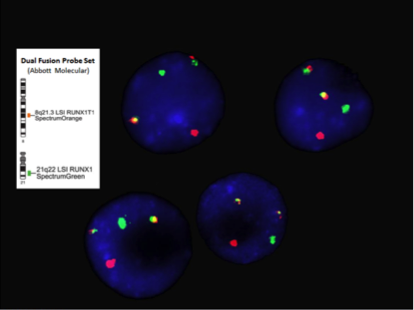
- Chromosome analysis, FISH, RT-PCR
- FISH or RT-PCR is needed for cytogenetically cryptic cases with typical blast morphology and immunophenotype
- RT-PCR is employed for minimal residual disease detection
Clinical Significance (Diagnosis, Prognosis and Therapeutic Implications)
Diagnostic:
- The presence of t(8;21)(q22;q22) is diagnostic of a cytogenetic subtype of acute myeloid leukemia with maturation in the neutrophilic lineage.
- When t(8;21)(q22;q22) is present the diagnosis of acute myeloid leukemia is made regardless of a blast count less than 20%.
Prognostic:
- t(8;21)(q22;q22) positive AML usually has a good response to chemotherapy and a high complete remission rate with long-term disease-free survival when treated with high dose cytarabine in the consolidation phase. Factors that appear to adversely affected prognosis include CD56 expression and KIT gene mutations. The prognostic significance of other accompanying gene mutations is under investigation.
Therapeutic target:
- t(8;21)(q22;q22) positive AML currently has no molecular target for drug therapy.
- Standard induction 7 + 3 chemotherapy regimen of cytarabine for 7 days plus an anthracycline or anthracenedione for 3 days. In first complete remission patients younger than 60 years without a KIT mutation are treated with high dose cytarabine consolidation therapy. If a KIT mutation is present, bone marrow transplant is performed in first complete remission[14].
- Recently homoharringtonine, aclarubicin and cytarabine as the first course of induction therapy has been found to be highly effective for acute myeloid leukemia with t(8;21)(q22;q22)[6].
Familial Forms
None
Other Information
None
Links
KIT mutations in AML COSMIC database
www.uptodate.com cytogenetics in AML
www.genenames.org RUNX1
www.genenames.org RUNX1T1
References
- ↑ Arber, Daniel A.; et al. (2016). "The 2016 revision to the World Health Organization classification of myeloid neoplasms and acute leukemia". Blood. 127 (20): 2391–2405. doi:10.1182/blood-2016-03-643544. ISSN 1528-0020. PMID 27069254.
- ↑ Sangle, Nikhil A.; et al. (2011). "Core-binding factor acute myeloid leukemia". Archives of Pathology & Laboratory Medicine. 135 (11): 1504–1509. doi:10.5858/arpa.2010-0482-RS. ISSN 1543-2165. PMID 22032582.
- ↑ Arber DA, et al., (2017). Acute myeloid leukaemia with recurrent genetic abnormalities, in World Health Organization Classification of Tumours of Haematopoietic and Lymphoid Tissues, Revised 4th edition. Swerdlow SH, Campo E, Harris NL, Jaffe ES, Pileri SA, Stein H, Thiele J, Arber DA, Hasserjian RP, Le Beau MM, Orazi A, and Siebert R, Editors. IARC Press: Lyon, France.
- ↑ Paschka, Peter; et al. (2006). "Adverse prognostic significance of KIT mutations in adult acute myeloid leukemia with inv(16) and t(8;21): a Cancer and Leukemia Group B Study". Journal of Clinical Oncology: Official Journal of the American Society of Clinical Oncology. 24 (24): 3904–3911. doi:10.1200/JCO.2006.06.9500. ISSN 1527-7755. PMID 16921041.
- ↑ Kim, Hee-Jin; et al. (2013). "KIT D816 mutation associates with adverse outcomes in core binding factor acute myeloid leukemia, especially in the subgroup with RUNX1/RUNX1T1 rearrangement". Annals of Hematology. 92 (2): 163–171. doi:10.1007/s00277-012-1580-5. ISSN 1432-0584. PMID 23053179.
- ↑ 6.0 6.1 Krauth, M.-T.; et al. (2014). "High number of additional genetic lesions in acute myeloid leukemia with t(8;21)/RUNX1-RUNX1T1: frequency and impact on clinical outcome". Leukemia. 28 (7): 1449–1458. doi:10.1038/leu.2014.4. ISSN 1476-5551. PMID 24402164.
- ↑ 7.0 7.1 Micol, Jean-Baptiste; et al. (2014). "Frequent ASXL2 mutations in acute myeloid leukemia patients with t(8;21)/RUNX1-RUNX1T1 chromosomal translocations". Blood. 124 (9): 1445–1449. doi:10.1182/blood-2014-04-571018. ISSN 1528-0020. PMC 4148766. PMID 24973361.
- ↑ 8.0 8.1 Hartmann, Luise; et al. (2016). "ZBTB7A mutations in acute myeloid leukaemia with t(8;21) translocation". Nature Communications. 7: 11733. doi:10.1038/ncomms11733. ISSN 2041-1723. PMC 4895769. PMID 27252013.
- ↑ Park, Sang Hyuk; et al. (2015). "Incidences and Prognostic Impact of c-KIT, WT1, CEBPA, and CBL Mutations, and Mutations Associated With Epigenetic Modification in Core Binding Factor Acute Myeloid Leukemia: A Multicenter Study in a Korean Population". Annals of Laboratory Medicine. 35 (3): 288–297. doi:10.3343/alm.2015.35.3.288. ISSN 2234-3814. PMC 4390696. PMID 25932436.
- ↑ Qin, Ya-Zhen; et al. (2016). "Low WT1 transcript levels at diagnosis predicted poor outcomes of acute myeloid leukemia patients with t(8;21) who received chemotherapy or allogeneic hematopoietic stem cell transplantation". Chinese Journal of Cancer. 35: 46. doi:10.1186/s40880-016-0110-6. ISSN 1944-446X. PMC 4873994. PMID 27197573.
- ↑ Speck, Nancy A.; et al. (2002). "Core-binding factors in haematopoiesis and leukaemia". Nature Reviews. Cancer. 2 (7): 502–513. doi:10.1038/nrc840. ISSN 1474-175X. PMID 12094236.
- ↑ Downing, James R. (2003). "The core-binding factor leukemias: lessons learned from murine models". Current Opinion in Genetics & Development. 13 (1): 48–54. doi:10.1016/s0959-437x(02)00018-7. ISSN 0959-437X. PMID 12573435.
- ↑ Marcucci, Guido (2006). "Core binding factor acute myeloid leukemia". Clinical Advances in Hematology & Oncology: H&O. 4 (5): 339–341. ISSN 1543-0790. PMID 16728942.
- ↑ Tallman, Martin S.; et al. (2019-06-01). "Acute Myeloid Leukemia, Version 3.2019, NCCN Clinical Practice Guidelines in Oncology". Journal of the National Comprehensive Cancer Network: JNCCN. 17 (6): 721–749. doi:10.6004/jnccn.2019.0028. ISSN 1540-1413. PMID 31200351.
Notes
*Primary authors will typically be those that initially create and complete the content of a page. If a subsequent user modifies the content and feels the effort put forth is of high enough significance to warrant listing in the authorship section, please contact the CCGA coordinators (contact information provided on the homepage). Additional global feedback or concerns are also welcome.
Edited by: Fabiola Quintero-Rivera 7/21/2018