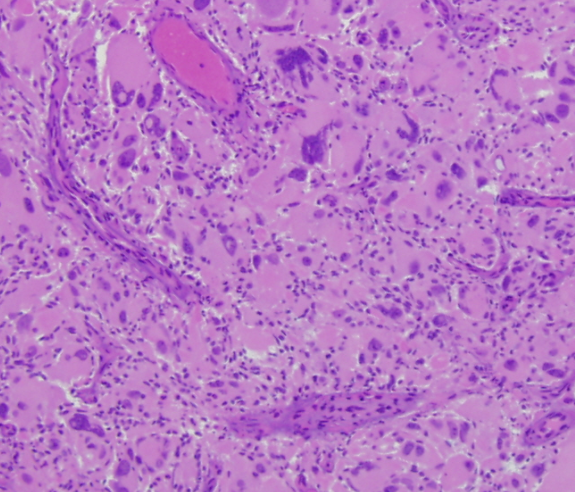
Giant cell glioblastoma
Primary Author(s)*
Jay Alden, DO
Cancer Category/Type
- Diffuse astrocytic and olidodendroglial tumors
Cancer Sub-Classification / Subtype
Glioblastoma, IDH-wildtype (IDH-wt)
Definition / Description of Disease
- Rare histologic variant of IDH-wt glioblastoma [1]
- Large, multinucleate giant cells with occasional abundant reticuln network [1]
Synonyms / Terminology
Epidemiology / Prevalence
- Constitute <1% of glioblastomas [2]
- May be more common in pediatric population [3]
- Mean age 51 years [3]
Clinical Features
- No evidence of a precursor lesion [1]
- Presenting symptoms similar to IDH-wt glioblastoma
- Radiographically and grossly circumscribed borders, may be mistaken for other neoplastic or non-neoplastic lesions [4]
Sites of Involvement
Morphologic Features
- Bizarre, multi-nucleate giant cells with atypical mitotic figures [5]
- Cells may contain numerous nuclei [1]
- Palisading and ischemic necrosis [1]
- Pseudo-rosette like perivascular tumor cell concentration [1]
Immunophenotype
| Finding | Marker |
|---|---|
| Positive (variable level) | GFAP [6] |
| Positive (subset) | P53 [7] [6] |
| Negative (universal) | neuN [8] |
Chromosomal Rearrangements (Gene Fusions)
Put your text here and/or fill in the table
| Chromosomal Rearrangement | Genes in Fusion (5’ or 3’ Segments) | Pathogenic Derivative | Prevalence |
|---|---|---|---|
| EXAMPLE t(9;22)(q34;q11.2) | EXAMPLE 3'ABL1 / 5'BCR | EXAMPLE der(22) | EXAMPLE 5% |
| EXAMPLE t(8;21)(q22;q22) | EXAMPLE 5'RUNX1 / 3'RUNXT1 | EXAMPLE der(8) | EXAMPLE 5% |
Characteristic Chromosomal Aberrations / Patterns
Put your text here
Genomic Gain/Loss/LOH
Put your text here and/or fill in the table
| Chromosome Number | Gain/Loss/Amp/LOH | Region |
|---|---|---|
| EXAMPLE 8 | EXAMPLE Gain | EXAMPLE chr8:0-1000000 |
| EXAMPLE 7 | EXAMPLE Loss | EXAMPLE chr7:0-1000000 |
Gene Mutations (SNV/INDEL)
Put your text here and/or fill in the tables
| Gene | Mutation | Oncogene/Tumor Suppressor/Other | Presumed Mechanism (LOF/GOF/Other; Driver/Passenger) | Prevalence (COSMIC/TCGA/Other) |
|---|---|---|---|---|
| EXAMPLE TP53 | EXAMPLE R273H | EXAMPLE Tumor Suppressor | EXAMPLE LOF | EXAMPLE 20% |
Other Mutations
| Type | Gene/Region/Other |
|---|---|
| Concomitant Mutations | EXAMPLE IDH1 R123H |
| Secondary Mutations | EXAMPLE Trisomy 7 |
| Mutually Exclusive | EXAMPLE EGFR Amplification |
Epigenomics (Methylation)
Put your text here
Genes and Main Pathways Involved
Put your text here
Diagnostic Testing Methods
Put your text here
Clinical Significance (Diagnosis, Prognosis and Therapeutic Implications)
Put your text here
Familial Forms
Put your text here
Other Information
Put your text here
Links
Put your links here
References
EXAMPLE Book
- Arber DA, et al., (2008). Acute myeloid leukaemia with recurrent genetic abnormalities, in World Health Organization Classification of Tumours of Haematopoietic and Lymphoid Tissues, 4thedition.Swerdlow SH, Campo E, Harris NL, Jaffe ES, Pileri SA, Stein H, Thiele J, Vardiman JW, Editors. IARC Press: Lyon, France, p117-118.
EXAMPLE Journal Article
- Li Y, et al., (2001). Fusion of two novel genes, RBM15 and MKL1, in the t(1;22)(p13;q13) of acute megakaryoblastic leukemia. Nat Genet 28:220-221, PMID 11431691.
Notes
*Primary authors will typically be those that initially create and complete the content of a page. If a subsequent user modifies the content and feels the effort put forth is of high enough significance to warrant listing in the authorship section, please contact the CCGA coordinators (contact information provided on the homepage). Additional global feedback or concerns are also welcome.
- ↑ 1.0 1.1 1.2 1.3 1.4 1.5 WHO Classification of Tumours of the Central Nervous System, 4th ed, Louis DN, Ohgaki H, Wiestler OD, Cavenee WK (Eds), IARC, Lyon 2016. Pg 46-47
- ↑ 2.0 2.1 Ortega, Alicia; et al. (2014). "Treatment and survival of patients harboring histological variants of glioblastoma". Journal of Clinical Neuroscience: Official Journal of the Neurosurgical Society of Australasia. 21 (10): 1709–1713. doi:10.1016/j.jocn.2014.05.003. ISSN 1532-2653. PMID 24980627.
- ↑ 3.0 3.1 3.2 Kozak, Kevin R.; et al. (2009). "Giant cell glioblastoma: a glioblastoma subtype with distinct epidemiology and superior prognosis". Neuro-Oncology. 11 (6): 833–841. doi:10.1215/15228517-2008-123. ISSN 1523-5866. PMC 2802403. PMID 19332771.
- ↑ Turner, Ryan; et al. (2018). "Imaging findings in the progression of a giant cell glioblastoma". Radiology Case Reports. 13 (5): 1007–1011. doi:10.1016/j.radcr.2018.07.010. ISSN 1930-0433. PMC 6097408. PMID 30128062.
- ↑ Margetts, J. C.; et al. (1989). "Giant-celled glioblastoma of brain. A clinico-pathological and radiological study of ten cases (including immunohistochemistry and ultrastructure)". Cancer. 63 (3): 524–531. doi:10.1002/1097-0142(19890201)63:33.0.co;2-d. ISSN 0008-543X. PMID 2912529.
- ↑ 6.0 6.1 Katoh, M.; et al. (1995). "Immunohistochemical analysis of giant cell glioblastoma". Pathology International. 45 (4): 275–282. doi:10.1111/j.1440-1827.1995.tb03456.x. ISSN 1320-5463. PMID 7550996.
- ↑ Peraud, A.; et al. (1997). "p53 mutations versus EGF receptor expression in giant cell glioblastomas". Journal of Neuropathology and Experimental Neurology. 56 (11): 1236–1241. doi:10.1097/00005072-199711000-00008. ISSN 0022-3069. PMID 9370234.
- ↑ Martinez-Diaz, Hilda; et al. (2003). "Giant cell glioblastoma and pleomorphic xanthoastrocytoma show different immunohistochemical profiles for neuronal antigens and p53 but share reactivity for class III beta-tubulin". Archives of Pathology & Laboratory Medicine. 127 (9): 1187–1191. doi:10.1043/1543-2165(2003)1272.0.CO;2. ISSN 1543-2165. PMID 12946225.