Acute erythroid leukaemia
Haematolymphoid Tumours (5th ed.)
| This page is under construction |
editHAEM5 Conversion NotesThis page was converted to the new template on 2023-11-03. The original page can be found at HAEM4:Pure Erythroid Leukemia.Note: need to check if content remains the same
Primary Author(s)*
Ashwini Yenamandra PhD FACMG
Cancer Category/Type
Cancer Sub-Classification / Subtype
Pure Erythroid Leukemia (PEL) is now the only type of Acute Erythroid Leukemia (AEL).
Definition / Description of Disease
In the 2008 WHO classification, Acute Erythroid leukemia (AEL) was classified into two subtypes: Erythroleukemia (erythroid/myeloid) and Pure Erythroid Leukemia (PEL). However, in the 2016 WHO update, Erythroleukemia was merged into myelodysplastic syndrome, while PEL is now the only type of AEL[1][2][3][4][5][6][7][8][9][10][11][12]. PEL is a distinct entity in the World Health Organization (WHO) classification system within the section of Acute Myeloid Leukemia (AML), Not Otherwise Specified. PEL is a rare form of acute leukemia with an aggressive clinical course and is characterized by an uncontrolled proliferation of immature erythroid precursors (proerythroblastic or undifferentiated)[1][2][3][4][5][6][7][8][9][10][11][12].
Synonyms / Terminology
Also known as AML-M6b and Di Guglielmo syndrome due to the recognition of the work of Di Guglielmo[1][2].
Epidemiology / Prevalence
PEL is extremely rare with a small number of reported cases, accounting for 3-5% of AML cases[1][2][10]. Median survival is usually three months[12].
Clinical Features
Put your text here and fill in the table (Instruction: Can include references in the table)
| Signs and Symptoms | EXAMPLE Asymptomatic (incidental finding on complete blood counts)
EXAMPLE B-symptoms (weight loss, fever, night sweats) EXAMPLE Fatigue EXAMPLE Lymphadenopathy (uncommon) |
| Laboratory Findings | EXAMPLE Cytopenias
EXAMPLE Lymphocytosis (low level) |
editv4:Clinical FeaturesThe content below was from the old template. Please incorporate above.PEL has an aggressive clinical course with neoplastic proliferation of immature erythroid precursor (proerythroblastic or undifferentiated) cells. Average survival rate is three months[1][10]. PEL is characterized by neoplastic proliferation composed of >80% immature erythroid precursors of which proerythroblasts constitute ≥30%[12]. Clinical features include profound anemia, circulating erythroblasts, pancytopenia, extensive bone marrow involvement, fatigue, infections, weight loss, fever, night sweats, hemoglobin level under 10.0 g/dL, and thrombocytopenia[1][10].
Sites of Involvement
Bone marrow, Blood
Morphologic Features
PEL is characterized by medium to large erythroblasts with round nuclei, fine chromatin and one or more nucleoli (proerythroblast). Cytoplasm is deeply basophilic, often granular with demarcated vacuoles and are often Periodic-Acid-Schiff stain (PAS) positive. Blasts can be small and may resemble lymphoblasts[1]. Cells are usually negative for Myeloperoxidase (MPO) and Sudan Black (SBB). Bone marrow biopsy may have undifferentiated cells[1].
Immunophenotype
Differentiated PEL may express Glycophorin and hemoglobin A, absence of myeloperoxidase (MPO) and other myeloid markers[1]. Blasts are negative for HLA-Dr and CD34 but positive for CD117[1]. Immature forms can be negative for Glycophorin or weekly expressed. Positive for Carbonic anhydrase 1, Gero antobody against the Gerbich blood group or CD36 especially at earlier stages of differentiation. CD41 and CD61 are negative[1][12].
| Finding | Marker |
|---|---|
| Positive (universal) | Hemoglobin A, Glycophorin A, Spectrin, ABH blood group antigens, and HLA-DR |
| Positive (subset) | CD13, CD33, CD34, CD117 (KIT), and MPO, Gerbich blood group (Gero) antibody, carbonic anhydrase 1, and CD36, CD41 and CD61 |
| Negative (universal) | Myeloid-associated markers such as MPO,CD13,CD33,CD61, B and T Cell markers -CD10, CD19, CD79a, CD2, CD3, CD5, monocytic markers CD11c CD14
Megakaryoblastic markers: CD61, Others: CD34, anti-kappa, anti-lambda, CD45 |
| Negative (subset) | HLA-DR, CD34, Glycophorin A |
Chromosomal Rearrangements (Gene Fusions)
Put your text here and fill in the table
| Chromosomal Rearrangement | Genes in Fusion (5’ or 3’ Segments) | Pathogenic Derivative | Prevalence | Diagnostic Significance (Yes, No or Unknown) | Prognostic Significance (Yes, No or Unknown) | Therapeutic Significance (Yes, No or Unknown) | Notes |
|---|---|---|---|---|---|---|---|
| EXAMPLE t(9;22)(q34;q11.2) | EXAMPLE 3'ABL1 / 5'BCR | EXAMPLE der(22) | EXAMPLE 20% (COSMIC)
EXAMPLE 30% (add reference) |
Yes | No | Yes | EXAMPLE
The t(9;22) is diagnostic of CML in the appropriate morphology and clinical context (add reference). This fusion is responsive to targeted therapy such as Imatinib (Gleevec) (add reference). |
editv4:Chromosomal Rearrangements (Gene Fusions)The content below was from the old template. Please incorporate above.The genetic abnormalities that have been identified in PEL are similar to that of AML and MDS and consists of complex chromosomal abnormalities including -5/del(5q), -7/del(7q), +8 and/or RUNX1 and TP53 mutations[1]. Rearrangement of NFIA-CBFA2T3 with t(1;16)(p31;q24) and MYND8-RELA with t(11;20)(p11;q11) have been reported in rare cases[10]. A complex karyotype with 46,XY,der(5)del(5)(p15.1p15.1)t(5;12;7)(p15.1;p13;q32),der(7)t(5;12;7),der(12)del(12)p13p13)t(5;12;7),del(13)(q12q14) was reported in a two year old boy with PEL[11].
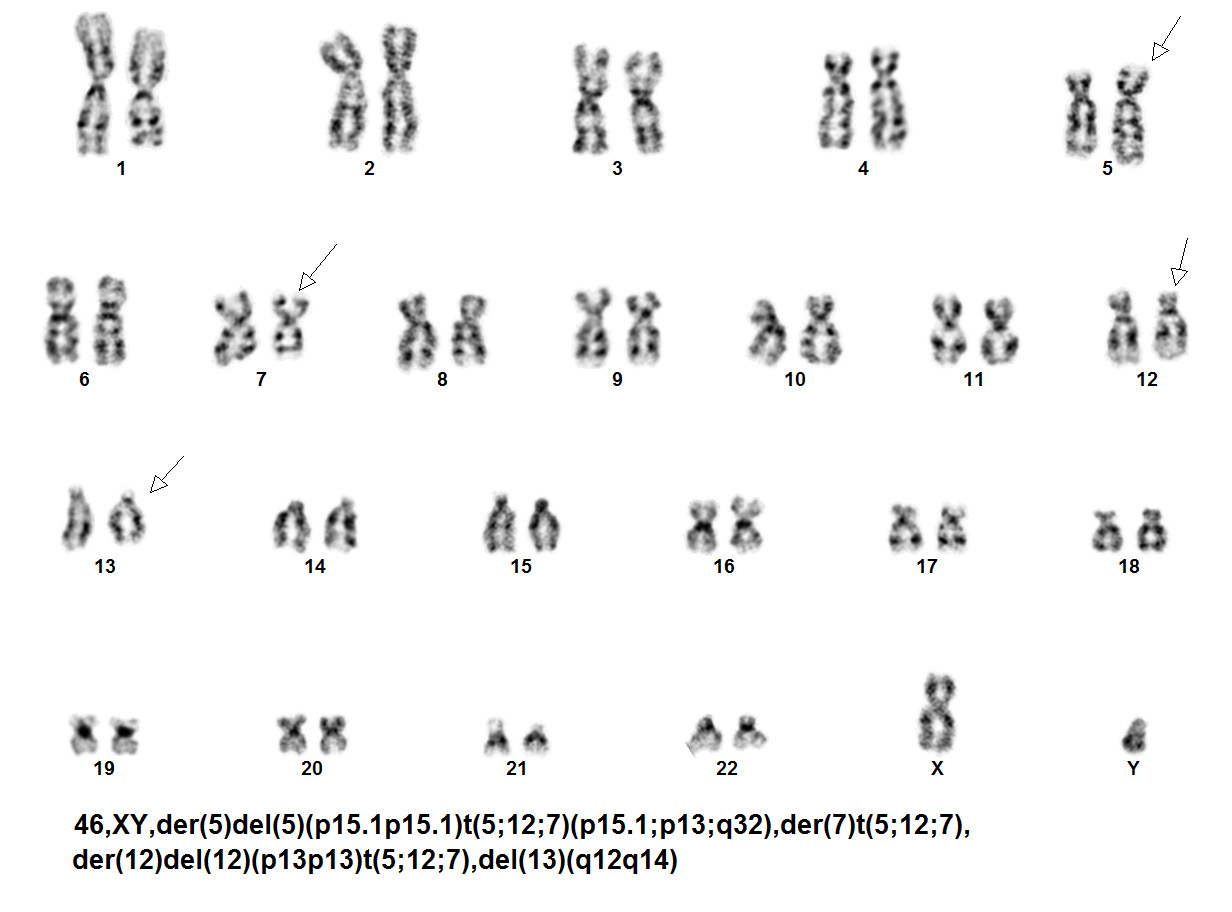 Karyotype of two year old boy with PEL[11].
Karyotype of two year old boy with PEL[11].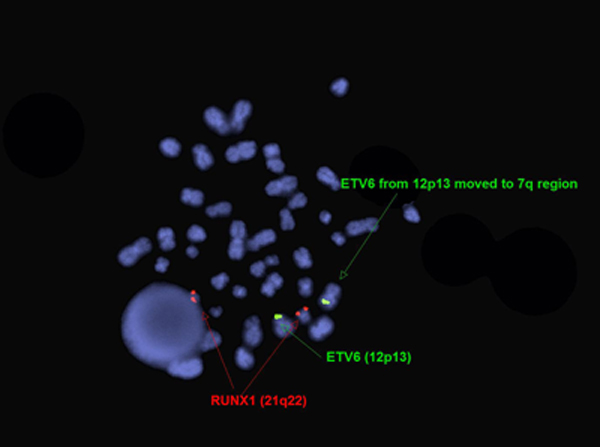 FISH for ETV6 and RUNX1 in two year old boy with PEL[11].
FISH for ETV6 and RUNX1 in two year old boy with PEL[11]. FISH for EGR1 and D5S23,D5S721 (5p-) in two year old boy with PEL[11].
FISH for EGR1 and D5S23,D5S721 (5p-) in two year old boy with PEL[11].
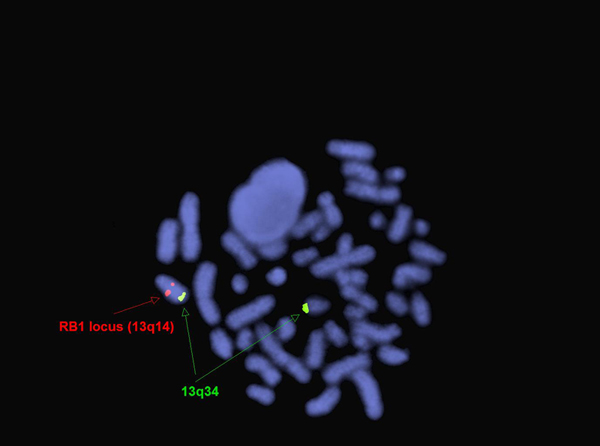 FISH for RB1 and 13q34 (13q deletion) in two year old boy with PEL[11].
FISH for RB1 and 13q34 (13q deletion) in two year old boy with PEL[11].
Chromosomal Rearrangement Genes in Fusion (5’ or 3’ Segments) Pathogenic Derivative Prevalence t(1;16)(p31;q24) 5’NFIA/ 3’CBFA2T3 der(16) Rare t(11;20)(p11;q11) 5’MYND8/ 3’RELA der(11) Rare
editv4:Clinical Significance (Diagnosis, Prognosis and Therapeutic Implications).Please incorporate this section into the relevant tables found in:
- Chromosomal Rearrangements (Gene Fusions)
- Individual Region Genomic Gain/Loss/LOH
- Characteristic Chromosomal Patterns
- Gene Mutations (SNV/INDEL)
PEL has rapid and aggressive clinical course. Patients with PEL are treated similar to other types of AML. Stem cell transplantation (SCT) may have an improvement in the outcome of the disease. No therapeutic agents for specific target pathways are currently available[3].
Individual Region Genomic Gain/Loss/LOH
Put your text here and fill in the table (Instructions: Includes aberrations not involving gene fusions. Can include references in the table. Can refer to CGC workgroup tables as linked on the homepage if applicable.)
| Chr # | Gain / Loss / Amp / LOH | Minimal Region Genomic Coordinates [Genome Build] | Minimal Region Cytoband | Diagnostic Significance (Yes, No or Unknown) | Prognostic Significance (Yes, No or Unknown) | Therapeutic Significance (Yes, No or Unknown) | Notes |
|---|---|---|---|---|---|---|---|
| EXAMPLE
7 |
EXAMPLE Loss | EXAMPLE
chr7:1- 159,335,973 [hg38] |
EXAMPLE
chr7 |
Yes | Yes | No | EXAMPLE
Presence of monosomy 7 (or 7q deletion) is sufficient for a diagnosis of AML with MDS-related changes when there is ≥20% blasts and no prior therapy (add reference). Monosomy 7/7q deletion is associated with a poor prognosis in AML (add reference). |
| EXAMPLE
8 |
EXAMPLE Gain | EXAMPLE
chr8:1-145,138,636 [hg38] |
EXAMPLE
chr8 |
No | No | No | EXAMPLE
Common recurrent secondary finding for t(8;21) (add reference). |
editv4:Genomic Gain/Loss/LOHThe content below was from the old template. Please incorporate above.Not Applicable
Chromosome Number Gain/Loss/Amp/LOH Region 5 Loss Whole chromosome or q-arm 7 Loss Whole chromosome or q-arm 8 Gain Whole chromosome 17 Loss P-arm
Characteristic Chromosomal Patterns
Put your text here (EXAMPLE PATTERNS: hyperdiploid; gain of odd number chromosomes including typically chromosome 1, 3, 5, 7, 11, and 17; co-deletion of 1p and 19q; complex karyotypes without characteristic genetic findings; chromothripsis)
| Chromosomal Pattern | Diagnostic Significance (Yes, No or Unknown) | Prognostic Significance (Yes, No or Unknown) | Therapeutic Significance (Yes, No or Unknown) | Notes |
|---|---|---|---|---|
| EXAMPLE
Co-deletion of 1p and 18q |
Yes | No | No | EXAMPLE:
See chromosomal rearrangements table as this pattern is due to an unbalanced derivative translocation associated with oligodendroglioma (add reference). |
editv4:Characteristic Chromosomal Aberrations / PatternsThe content below was from the old template. Please incorporate above.
Gene Mutations (SNV/INDEL)
Put your text here and fill in the table (Instructions: This table is not meant to be an exhaustive list; please include only genes/alterations that are recurrent and common as well either disease defining and/or clinically significant. Can include references in the table. For clinical significance, denote associations with FDA-approved therapy (not an extensive list of applicable drugs) and NCCN or other national guidelines if applicable; Can also refer to CGC workgroup tables as linked on the homepage if applicable as well as any high impact papers or reviews of gene mutations in this entity.)
| Gene; Genetic Alteration | Presumed Mechanism (Tumor Suppressor Gene [TSG] / Oncogene / Other) | Prevalence (COSMIC / TCGA / Other) | Concomitant Mutations | Mutually Exclusive Mutations | Diagnostic Significance (Yes, No or Unknown) | Prognostic Significance (Yes, No or Unknown) | Therapeutic Significance (Yes, No or Unknown) | Notes |
|---|---|---|---|---|---|---|---|---|
| EXAMPLE: TP53; Variable LOF mutations
EXAMPLE: EGFR; Exon 20 mutations EXAMPLE: BRAF; Activating mutations |
EXAMPLE: TSG | EXAMPLE: 20% (COSMIC)
EXAMPLE: 30% (add Reference) |
EXAMPLE: IDH1 R123H | EXAMPLE: EGFR amplification | EXAMPLE: Excludes hairy cell leukemia (HCL) (add reference).
|
Note: A more extensive list of mutations can be found in cBioportal (https://www.cbioportal.org/), COSMIC (https://cancer.sanger.ac.uk/cosmic), ICGC (https://dcc.icgc.org/) and/or other databases. When applicable, gene-specific pages within the CCGA site directly link to pertinent external content.
editv4:Gene Mutations (SNV/INDEL)The content below was from the old template. Please incorporate above.JAK2, FLT3, RAS, NPM1, and CEBPA mutations have been reported to be rare in PEL[10][11][12]. Intraclonal heterogeneity and founder mutations of TP53 were reported in 92% (11 out of 12 cases) while co-occurrence of TP53 mutation and deletion due to chromosome 17p abnormalities were detected in 73% of PEL cases[13].
Gene Mutation Oncogene/Tumor Suppressor/Other Presumed Mechanism (LOF/GOF/Other; Driver/Passenger) Prevalence (COSMIC/TCGA/Other) EXAMPLE TP53 EXAMPLE R273H EXAMPLE Tumor Suppressor EXAMPLE LOF EXAMPLE 20% Other Mutations
Type Gene/Region/Other Concomitant Mutations EXAMPLE IDH1 R123H Secondary Mutations EXAMPLE Trisomy 7 Mutually Exclusive EXAMPLE EGFR Amplification
Epigenomic Alterations
Not Applicable
Genes and Main Pathways Involved
Put your text here and fill in the table (Instructions: Can include references in the table.)
| Gene; Genetic Alteration | Pathway | Pathophysiologic Outcome |
|---|---|---|
| EXAMPLE: BRAF and MAP2K1; Activating mutations | EXAMPLE: MAPK signaling | EXAMPLE: Increased cell growth and proliferation |
| EXAMPLE: CDKN2A; Inactivating mutations | EXAMPLE: Cell cycle regulation | EXAMPLE: Unregulated cell division |
| EXAMPLE: KMT2C and ARID1A; Inactivating mutations | EXAMPLE: Histone modification, chromatin remodeling | EXAMPLE: Abnormal gene expression program |
editv4:Genes and Main Pathways InvolvedThe content below was from the old template. Please incorporate above.The molecular mechanism is not completely understood.
Genetic Diagnostic Testing Methods
Morphology and IHC.
Familial Forms
Not Applicable
Additional Information
Differential Diagnosis: PEL without morphological differentiation of erythroid maturation can be difficult to distinguish from megakaryoblastic leukemia (AML), ALL or lymphoma. The erythroid precursor immunophenotype helps in the diagnosis. Some cases can be complex with concurrent erythroid megakaryocytic involvement[1].
Links
References
(use the "Cite" icon at the top of the page) (Instructions: Add each reference into the text above by clicking on where you want to insert the reference, selecting the “Cite” icon at the top of the page, and using the “Automatic” tab option to search such as by PMID to select the reference to insert. The reference list in this section will be automatically generated and sorted. If a PMID is not available, such as for a book, please use the “Cite” icon, select “Manual” and then “Basic Form”, and include the entire reference.)
- ↑ 1.00 1.01 1.02 1.03 1.04 1.05 1.06 1.07 1.08 1.09 1.10 1.11 1.12 Arber DA, et al., (2008). World Health Organization Classification of Tumours of Haematopoietic and Lymphoid Tissues, 4th edition. Swerdlow SH, Campo E, Harris NL, Jaffe ES, Pileri SA, Stein H, Thiele J, Vardiman JW, Editors. IARC Press: Lyon, France, p135-136.
- ↑ 2.0 2.1 2.2 2.3 Qiu, Shaowei; et al. (2017). "An analysis of 97 previously diagnosed de novo adult acute erythroid leukemia patients following the 2016 revision to World Health Organization classification". BMC cancer. 17 (1): 534. doi:10.1186/s12885-017-3528-6. ISSN 1471-2407. PMC 5550989. PMID 28793875.
- ↑ 3.0 3.1 3.2 Zuo, Zhuang; et al. (2010). "Acute erythroid leukemia". Archives of Pathology & Laboratory Medicine. 134 (9): 1261–1270. doi:10.1043/2009-0350-RA.1. ISSN 1543-2165. PMID 20807044.
- ↑ 4.0 4.1 Arber, Daniel A.; et al. (2016). "The 2016 revision to the World Health Organization classification of myeloid neoplasms and acute leukemia". Blood. 127 (20): 2391–2405. doi:10.1182/blood-2016-03-643544. ISSN 1528-0020. PMID 27069254.
- ↑ 5.0 5.1 Zuo, Zhuang; et al. (2012). "Acute myeloid leukemia (AML) with erythroid predominance exhibits clinical and molecular characteristics that differ from other types of AML". PloS One. 7 (7): e41485. doi:10.1371/journal.pone.0041485. ISSN 1932-6203. PMC 3402404. PMID 22844482.
- ↑ 6.0 6.1 Grossmann, V.; et al. (2013). "Acute erythroid leukemia (AEL) can be separated into distinct prognostic subsets based on cytogenetic and molecular genetic characteristics". Leukemia. 27 (9): 1940–1943. doi:10.1038/leu.2013.144. ISSN 1476-5551. PMID 23648669.
- ↑ 7.0 7.1 Porwit, Anna; et al. (2011). "Acute myeloid leukemia with expanded erythropoiesis". Haematologica. 96 (9): 1241–1243. doi:10.3324/haematol.2011.050526. ISSN 1592-8721. PMC 3166090. PMID 21880638.
- ↑ 8.0 8.1 Hasserjian, Robert P.; et al. (2010). "Acute erythroid leukemia: a reassessment using criteria refined in the 2008 WHO classification". Blood. 115 (10): 1985–1992. doi:10.1182/blood-2009-09-243964. ISSN 1528-0020. PMC 2942006. PMID 20040759.
- ↑ 9.0 9.1 Wang, Sa A.; et al. (2015). "Acute Erythroleukemias, Acute Megakaryoblastic Leukemias, and Reactive Mimics: A Guide to a Number of Perplexing Entities". American Journal of Clinical Pathology. 144 (1): 44–60. doi:10.1309/AJCPRKYAT6EZQHC7. ISSN 1943-7722. PMID 26071461.
- ↑ 10.0 10.1 10.2 10.3 10.4 10.5 10.6 Wang, Wei; et al. (2017). "Pure erythroid leukemia". American Journal of Hematology. 92 (3): 292–296. doi:10.1002/ajh.24626. ISSN 1096-8652. PMID 28006859.
- ↑ 11.0 11.1 11.2 11.3 11.4 11.5 11.6 11.7 A, Yenamandra (2016). "Acute Erythroblastic Leukemia (AEL): A Rare Subset of De Novo AML with A Complex Rearrangement Involving ETV6 Locus and Loss of RB1 Locus". International Clinical Pathology Journal. 2 (2). doi:10.15406/icpjl.2016.02.00032.
- ↑ 12.0 12.1 12.2 12.3 12.4 12.5 Fs, Khan (2017). "Pure Erythroid Leukemia: The Sole Acute Erythroid Leukemia". International Journal of Bone Marrow Research. 1 (1): 001–005. doi:10.29328/journal.ijbmr.1001001.
- ↑ Montalban-Bravo, Guillermo; et al. (2017). "More than 1 TP53 abnormality is a dominant characteristic of pure erythroid leukemia". Blood. 129 (18): 2584–2587. doi:10.1182/blood-2016-11-749903. ISSN 1528-0020. PMC 5418636. PMID 28246192.
Notes
*Primary authors will typically be those that initially create and complete the content of a page. If a subsequent user modifies the content and feels the effort put forth is of high enough significance to warrant listing in the authorship section, please contact the CCGA coordinators (contact information provided on the homepage). Additional global feedback or concerns are also welcome. *Citation of this Page: “Acute erythroid leukaemia”. Compendium of Cancer Genome Aberrations (CCGA), Cancer Genomics Consortium (CGC), updated 11/3/2023, https://ccga.io/index.php/HAEM5:Acute_erythroid_leukaemia.